Why rainbows and oil slicks help to show the greenhouse effect
Posted on 3 February 2014 by MarkR
The greenhouse effect, where some atmospheric gases let through sunlight but absorb the infrared heat given off by Earth, can be understood in amazing detail by starting off with the spread of colours in a rainbow or on an oil slick. Greenhouse gases warm Earth’s surface by about 33 C and the carbon dioxide we’re releasing by (mostly) burning coal, oil and gas is seen by 97% of climate scientists to be causing most of the recent global warming.
This is a very unpopular message, and it has been attacked using loads of myths we’ve covered on Skeptical Science. One of the most ‘out there’ is that the greenhouse effect doesn’t exist, so carbon dioxide can’t cause global warming (e.g. quoted here).
These claims are not taken seriously by climate scientists, missile designers, laser physicists, astrophysicists or meteorologists. Even self-styled 'skeptical' blogger Anthony Watts doesn't buy it.
The greenhouse effect is measured by instruments like this one every single day:

Figure 1: A pyrgeometer, a device for measuring infrared light coming down from the sky. Even at night it measures infrared, thanks to the greenhouse effect (image: Wiki).
And if these greenhouse skeptics were right, then lasers and heat-seeking missiles wouldn’t work like they do, and we couldn’t have measured the afterglow of the Big Bang either.
But what about rainbows and oil slicks? They reveal something very important about light: we can split it up into different colours. It even comes in colours we can’t see like infrared or ultraviolet. Greenhouse gases in the sky shine down infrared light on us day and night, keeping us warm. And more greenhouse gases equals more warmth.

Figure 2: Takakkaw Falls rainbow in the misty spray from the waterfall. Rainbows show that light is made of many colours, and we can split light and analyse these colours separately. This is a key technique that has identified the greenhouse effect (stunning image courtesy of Wiki).
Just as a rainbow or oil slick splits up light into different colours we can see, from red to violet, we have tools that split up infrared light to give an infrared spectrum. We can measure the brightness of each infrared colour, and find out an amazing amount about the greenhouse effect and the gases in the atmosphere.
The colour of light from a gas depends on exactly what that gas is, the familiar glow of hot sodium vapour is the dull orange spilled by street lamps.

Figure 3: A lamp, where the orange colour of its glow reveals that the gas inside contains sodium vapour (Wiki).
In the atmosphere, each gas has its own spectrum and some gases like water vapour and CO2 have an infrared glow that makes them important for the greenhouse effect. And this has been measured over and over again using the full spectrum.
According to the greenhouse skeptics this is impossible: there can’t be radiation coming down from the atmosphere and heating us up. They have written blog posts and even made their own magazines to publish articles on this, but have failed to publish in expert-reviewed scientific journals.
The sort of work that has got into these expert journals includes Tjemkes et al. (2003), where they flew aircraft off the US Eastern Seaboard and near Ascension Island and measured the infrared spectrum in great detail. They then used standard physics, including a computer code developed by the US Department of Defence for guiding heat-seeking missiles.
For almost every bit of the infrared spectrum, the physics predicted the measurement to an accuracy of better than 1%.
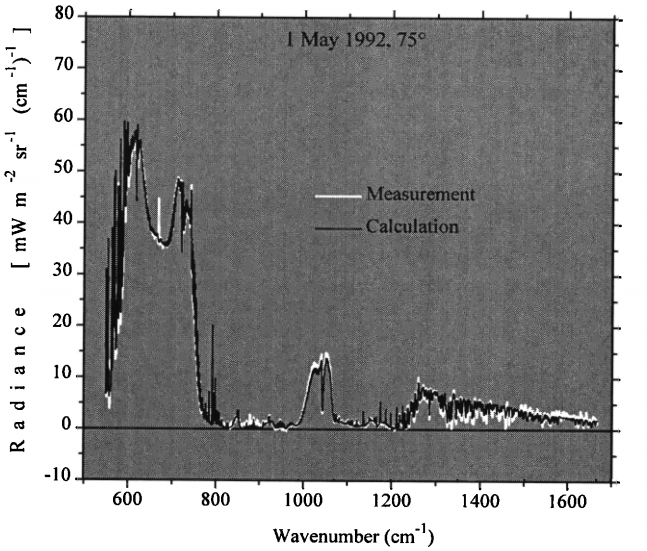
Figure 4: One example of comparing a measured spectrum with physics calculations. The bottom axis shows the 'colour' of the light and the side axis shows how bright the light is at that colour. The measurements (white) almost exactly match what is calculated (black). Graph modified from Walden et al. (1998).
Physics works brilliantly, and it works across the entire spectrum for every ‘colour’ of infrared, whether you’re measuring the glow of the sky as it shines down on the Antarctica plateau (Walden et al. (1998)) or using the IASI instrument strapped to a satellite as it flies over Sodankylä in Finland (Calbet et al. (2011)).
For almost every colour of infrared, standard physics makes an almost-perfect prediction. Modern research is filling in the details and won’t change the results by more than a few percent.
Science makes ‘models’ which try to predict how the real world works, and the amazingly precise models we have today have been tested over and over again, and that’s why the greenhouse effect is accepted by scientists worldwide.
Those who deny the greenhouse effect have written reams of blog posts and magazine articles full of bluster. They haven’t produced a model that explains any of these measurements.
These models are also a key part of how we know that we are heating up Earth. Adding greenhouse gases (like CO2 and methane) traps more heat in the atmosphere (Myhre et al. (1998)). This change has been measured by satellites which split the spectrum (e.g. Harries et al. (2001), Chen et al. (2007), Chapman et al. (2013)). The first of those studies has led to a graph we've often posted on Skeptical Science, showing the change in the amount of infrared light escaping from Earth depending on its colour. Thanks to this, we know that our greenhouse gases are trapping enough heat to be responsible for most of the recent global warming, and that there's a lot more warming to come.
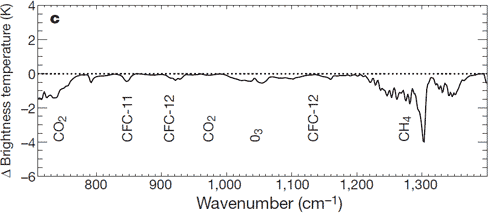
Figure 5: Direct measurement of the man-made greenhouse effect getting stronger, thanks to carbon dioxide (CO2) and methane (CH4). This is the change in how much energy is escaping Earth at different 'colours' from 1970 to 1996. Absorption by carbon dioxide and methane means that the satellites see less at those colours than they did in the past: this extra heat warms the Earth. Image from Harries et al. (2001)































 Arguments
Arguments






























Great to see some clear evidence that answers the common 'skeptic' demand for empirical evidence of enhanced greenhouse warming. Though I am not myself a climate scientist (artist here, heh heh) I have been drawn into the arguments quite a bit in media commentary, and have cited the example of heat-seeking missile technology a number of times. Suppose I should get some proper references for the actual work in that field (maybe some of it is classified info, not sure) but it is fairly well known that the early development of such technology had to work out instrumental errors due to the heat masking properties of CO2 and other GHGs, and to re-calibrate such guidance systems in order to make them work properly.
I can never resist the zinger which most effectively closes this Q.E.D. response to the 'no greenhouse effect' claims: should these folks find themselves up in the air with a heat-seeking missile on their tail, they can bet their flaming backsides, its aim is true!
Like Andrew Mclaren, I am not a scientist (poet here ...), but I agree that this is a useful article to be able to link to. Sadly, the committed denialospheroids will dismiss it as just more spin and claim it has been 'widely discredited' (though so far they have been curiously unable to point to the scientific papers discrediting anything about which they make this claim). Neverthless, I will gladly add this arrow to my quiver of links to post in rebuttal of denialist comments on other sites I haunt, such as The Conversation, on the basis that any ambivalent people reading such a thread of comments need to be pointed to reliable information, to counter the ridiculous denialospheroid claims.
"it has been attached using loads of"
perhaps "attacked" ???
Damn it bjchip, I'm a scientist not a linguist. Thanks for the catch, correction made.
There have been so many articles devoted to explaining the greenhouse effect. What I hoped to get across here was how thsoe who don't like it have completely failed to present any explanation for the spectral measurements. Aside from denying that these measurements exist or are possible.
Given that, as already said, it's rather difficult to write anything that will convince a skeptic, I keep on mulling whether it's possible to build some sort of `greenhouse room' that could be used in science museums to illustrate the effect. Can someone actually able to do the science sums tell me if this is nonsense? I have a feeling you'd need lots of other factors like cold enough co2...
Would it be possible to have: (a) the bottom half of a 20 foot high ceiling room open to people via a door (with air controls, see below); (b) the top half a sealed container with two separate compartments, one containing the same air mix as the room itself, the other pure co2? Each part could be moved over the room, hiding the other, with some powerful light source above it. The floor of the room could be something that's reflecting back more of the IR. (You'd also need to carefully define the in-out flow of air to the room itself so you're not suffocating people while also allowing for a predictable change in temps as the IR bouncing back heats things up).
What would it take for that to show a measurable effect? Given I don't much know what I'm talking about, are there are other similar room setups that might allow people to directly experience the effect of a CO2 blanket on the air temp of the room they're in? (If I were being cruel, I'd quite like one where skeptics who claim no such effect exist could be put in one where the temp could be raised to 60C this way...)
Of course, I suppose if you build such a thing, skeptics would simply say "the atmosphere's completely different, don't be silly". As a general rule, though, it'd be targetting sensible waverers to innoculate them against FUD, not skeptics themselves, who are beyond our aid I suspect.
Here's another "proof" that's very simple: weknow the greenhouse effect is there because we can survive at night!
If the ground temperature at sundown in summer is 60 F (about 15 C or 288 Kelvin), then by the Stefan-Boltzmann Law it is emitting sigma * T^4 = 6.67e-8 * (288)^4 = 390 Watts per square meter. If this cooling were felt through a 10 cm thick layer of soil, the ground temperature would cool by 75 Kelvin over 8 hours of darkness, reaching -60 C (-78 F) by morning.
Luckily for us CO2 and water vapor molecules in the air emit infrared radiation downward at over 300 Watts per square meter, so we can survive night on Earth!
To be fair, only the really wacky fringe actually deny that CO2 emits heat. But I have actually met a few, and of course the original post links to some of these claims.
It's not just physics that's studied infrared (IR).
In chemistry, the interaction between IR and matter is studied in great detail. Today, IR spectroscopy is routine in any chemistry lab. It helps identify unknown compounds, because we know which IR absorption peak shapes (similar to Figure 4) correspond to many types of bonds between specific atoms. Even a C student in undergrad organic chemistry can tell an alcohol from an aldehyde by glancing at the IR signatures.
How would those who deny the basic physics of IR-matter interaction propose we identify chemical unknowns? Look them up in a scratch and sniff index?
Doug, the link to Met Office's world IR satellite imagery might help to make the greenhouse/IR link clear to people. Looking at it in this season, Australia provides a good example of hot land, and overlying cloud tops being much cooler due to the warmth being trapped at lower levels due to the greenhouse action of water vapor.
http://www.metoffice.gov.uk/weather/satellite/ shows the world, but you can click to select parts of the planet, and look at differences between daytime and nighttime, too.
bf @8, clouds are (depending on thickness) almost perfect black or grey bodies in the IR spectrum. That is, they absorb and radiate equally at all IR wavelengths. So also does the ground and and water at the surface. In contrast, CO2 and H2O in gaseous form only absorb and radiate at certain wavelengths. Satellites designed to observe clouds will almost certainly have instruments tuned to wavelengths at which CO2 and gaseous H2O do not absorb. That is so that clouds at even low levels of the atmosphere can be detected. Consequently, while the satellite imagary of clouds you link to is impressive evidence that the upper troposphere is cooler than the surface, it is not evidence of the greenhouse effect. Of course, evidence that altitude cools is abundant, the most obvious example being snow capped mountains.
All else being equal, the greenhouse effect would actually cool the atmosphere with altitutude faster than it does, but convection results in a much warmer upper troposphere. Howeve, convection by itself would also cool with altitude. The reason is simple conservation of energy. As gas particles rise higher in the sky, they gain gravitational potential energy. Because of conservation of energy, that energy gained must come from somewhere else, and typically comes from the kinetic energy - that is the energy of motion plus energy of vibration - of the molecules. If the rising air column contains water vapour, the water vapour will precipitate out as it cools, also providing energy, and therefore allowing a slower loss of temperature with altitude.
Below the tropopause, vertical transfer of energy via convection dominates over vertical transfer by radiation. The result is that the loss of temperature with altitude is governed by convection and the loss of latent heat as water vapour precipitates, such that the loss of temperature with altitude would be the same even with no greenhouse effect.
Thanks, for the correction, Tom.
I'd read David Randall's "Atmosphere, Clouds and Climate" last year, but clearly need another go-through for it to begin to stick. Maybe this time I'll work through some of the equations instead of just filing them under "more greek symbols".
Super article, Mark. Thanks!
...there can’t be radiation coming down from the atmosphere and heating us up.
Leaving aside Mark's lovely explanation, what I've never seen fully developed by skeptics (dismissives; whatever) making claims about the destination and ultimate fate of radiation is how photons know where they're supposed to be going. If somehow radiation avoids going from a cooler body to a wamer body, that behavior would require superluminal information transfer, a mechanism for photons to sort and choose their destinations, etc. A whole pile of "somehow" is left unaddressed.
This article mischaracterizes this out of the many skeptic arguments as..."One of the most ‘out there’ is that the greenhouse effect doesn’t exist..." That is a fringe argument which goes against basic physics. It is not "one of the most out there" as characterized. As you note, not even Anthony Watts accepts this argument. Goes in the same bucket as fringe arguments such as forecasts of the arctic being ice free in 2013.
Something else is when Skeptics claim that 300 parts per MILLION is just too low a concentration to have any affect on 'darkening' the atmosphere to infrared light. Why, that's "next to NOTHING"! To that, I point out this ad:
http://www.poolcenter.com/p/party-pool-swimming-pool-color-dye
Yes, a mere 8oz of this dye will turn your crystal clear 20,000 gallon swimming pool black to visible radiation, twice over!
That's (half a cup/20,000 gallons)*(1gallon/16 cups) = 1.6 ppm. Oops, "Nothing" has magically become "Something"!
joeygoze @12 said: "[Disbelieving the greenhouse effect] Goes in the same bucket as fringe arguments such as forecasts of the arctic being ice free in 2013" You can't seriously believe these are in the same bucket! The former is a refutation of basic Physics now two hundred years old. The latter is a forecast consistent with the last 30 year trend. There's a one minute stretch of this video that starts at 0:50, which shows the month by month trend in Arctic ice since the 1980s. Tell me you can look at that trend and NOT expect an ice free Arctic summer in the next decade or so!
https://www.youtube.com/watch?v=ZYaubXBfVqo
I am referring to forecasts made in 2007, 2008 and 2009 predicting an ice free arctic in 2013. Clearly over the top and did not come true.
It's almost certainly far less than 1.6 ppm, ubrew, since the dye itself is almost certainly itself a solution.
ubrew12 @13 and Jim. I sometimes counter the "but it's such a tiny amount" argument by suggesting the person posting it to try the same when a cop is giving them a roadside breathalyzer test. "But officer, that tiny fraction of alchohol by volume just CAN'T have affected my driving."
joeygoze@15,
It needs to be clarified, that "forecasts made in 2007, 2008 and 2009 predicting an ice free arctic in 2013" were not any forcasts but simple & primitive distortion of scientific literature by deniers.
Wieslaw Maslowski predicted back in 2007 (before we saw the Sept 2007 minimum) "nearly ice free arctic" in 2016+/-3years, if the trend (as pointed by ubrew12@14) continues.
Deniers took Maslowski sentence, removed the "if the trend continues" clause, removed uncertainty, instead picking up the lower bound of uncertainty value (2013) and estaqblished it as the abosolutely certain, central value, thus creating the bogus, moronic slogan you're refering to.
So this "argument" by deniers is so silly that no scientist (inc maslowski) bothers to even listen, but we must debunk such arguments over and over because simple slogans "stick hard" to many minds. And unfortunately, many of those affected minds are supposed to take responsibility to reverse AGW (e.g. US Congress) but stick their heads in the sand.
There is an important need to debunk such slogans with basic science, and this article does good job with several of them related to IR.
joeygoze #15 different bucket. I've computed with virtual certainty that it's more than 5 decimal orders of magnitude more incompetent to disbelieve a solid bit of physics, that school kids are demonstrating in videos, than to predict 2013 as the exact year in which the arctic will be ice free. Nonetheless, please provide a link to some of the persons who said that so that we can ponder it.
chriskoz@18
If you want to argue on who is parsing words, the predictions made in 2007, 2008, and 2009 I was taking from Al Gore so if he failed to use the words "if the trend continues", I would correct your statement to say it was "simple and primative distortion of the scientific literature by alarmists" That "moronic slogan" was repeated by Al Gore, John Kerry and even as late as March of 2013, Paul Beckwith from the Sierra Club when he wrote "“For the record—I do not think that any sea ice will survive this summer (2013). An event unprecedented in human history is today, this very moment, transpiring in the Arctic Ocean." Italics is added to refer to the year he is speaking about.
My issue with the above article is the characterization that the denial of a greenhouse effect existing at all is mainstream argument. That is simply not true. No one from any side of this discussion in the mainstream is arguing that CO2 can not trap heat or that No greenhouse effect exists. If there was NO greenhouse effect, then the Earth would be very inhospitable place to live.
[JH] Please doument your source of information about the statements supposedly made by Al Gore, John Kerry, and Paul Beckwith.
BTW, Al Gore, John Kerry, and Paul Beckwith are not climate scientists.
Denier inflation alert: Given the stunning retreat of summer Arctic Ice in the last 20 years, why is the 'fact' that ice didn't disappear COMPLETELY in 2013 some kind of 'proof' that Global Warming is a hoax? Why, if the five hottest years in known history occurred since the turn of this century (according to Cowtan & Way's analysis), yet temperatures didn't increase as much as they did between 1978 and 1998, is this proof that Global Warming is a hoax?
The value of 'Doubt is our Product' denialism is the assumption that 'they' own the goalposts, and will move them whereever they want. Because, don't ya know, they are just THAT kind of sticklers for perfection. All I can say is: I want THAT job!
I can't believe the number of deniers who have told me the Climate Models are broken. Who have no Climate Models, and can't point to any, despite being backed by the most profitable industry in the history of capitalism, that models EVERYTHING, from oil tanker designs, to oil fields, to chemical refineries, to pipelines, and for which Climate Change as an existential threat to its profitability. No competing Climate models of any kind.
This reminds me of Graffiti taggers. Someone else builds a bridge and people say 'its beautiful'. Then the tagger paints the bridge with his favorite swear word and points out 'no, its not'. And then, amazingly, some people start to agree with him...
#12 joeygoze: it is one of the craziest IMO, but I think this discussion is worthwhile because people who start looking at climate but don't have a strong physics background (which is most people?) could easily find one of the Principia Scientific sites while googling for the greenhouse effect and fall for it.
You're right in saying that it's completely physically baseless, but the same goes for many of the more common myths that you see on the more 'mainstream' pseudoskeptic sites. 'Warming has paused' contradicts statistics. 'The deep oceans can't be warming without heating up the upper layers' also contradicts basic physics. So does 'CO2 is saturated' or 'CO2 is only a tiny portion of the atmosphere so it can't cause warming'.
So I don't think that 'the pseudoskeptic argument is completely physically unrealistic' is a good reason to avoid posting about it. :)
joeygoze@20
That is simply not true. Obvious examples of influential peoples (who have thousands of followers) who still deny GFE are: james inhofe in US Congress, Cuccinelli in Senate of VA, Tony Abbott - AUS PM, who said that climate science is "crap" and that CO2 is invisible substance.
Therefore an article as this is needed to debunk Tony Abbott's taking point, because Tony does implicitly deny the existence of GHE here and his argument dismissing GHE is indeed his "mainstream argument". Which proves that your statement above is not true.
joeygoze@20,
Your "alarmists" may also be considered "science deniers", because they did not check the scientific source before pronouncing the slogan. Or, more likely, they may have fallen victims of the slogan, because they did not have time/will to verify it. They, like the deniers obstructing action against AGW I refered to earlier, also need education so that they also understand the problem of AGW and do not raise 'false alarm'.
Back to the subject of your objection re this article. I may skid onto thin ice of comment policy however cannot help but ask, what's the motivation of your objection? I've already proven you to be wrong on your assertion that "No one from any side of this discussion in the mainstream is arguing that CO2 can not trap heat". You understand that basic science and you don't need this article. Good for you. But do you want those poeple who are not as fortunate not to learn the basic climate science stuff?
Moderator:
Paul Beckwith - http://www.sierraclub.ca/en/AdultDiscussionPlease
John Kerry - ""It is already upon us and its effects are being felt worldwide, right now," he wrote. "Scientists project that the Arctic will be ice-free in the summer of 2013. Not in 2050, but four years from now. Make no mistake: catastrophic climate change represents a threat to human security, global stability, and — yes — even to American national security." http://www.politifact.com/truth-o-meter/statements/2009/sep/02/john-kerry/kerry-claims-arctic-will-be-ice-free-2013/
Al Gore - "Last September 21 (2007), as the Northern Hemisphere tilted away from the sun, scientists reported with unprecedented distress that the North Polar ice cap is "falling off a cliff." One study estimated that it could be completely gone during summer in less than 22 years. Another new study, to be presented by U.S. Navy researchers later this week, warns it could happen in as little as 7 years."
So correction on the Al Gore assertion, if it happened in as little as 7 years, would be the 2014 minimum would be ice free, not 2013
[JH] Thank you for providing the references. Why are the statements of these three non-scientists important and relevant?
All: joeygoze's most recent comment was a moderation complaint and therefore was deleted.
The statements of the 3 non-scientists are as relevant as Sarah Palin or Jame Inhofe statements raised by Composer99. For a scientific discussion, those individuals are not relevant at all. Saying all those public figures do not represent mainstream scientific discussion.
As far as the accusation of "tone trolling" or "poisoning the well", no comment. That is an accusation against me, not my argument. Just to be clear, I agree with 99% in the above article, the sciecne of a greenhouse effect that warms the planet is clear basic physics. The assertion that a majority of people that question man's contribution to the greenhouse effect also do not believe in a greenhouse effect at ALL is what I was talking about. That is not a "mainstream" scientific position. Along the same lines, I do not accept everything Al Gore says as the mainstream position of the majority of climate scientists. i.e, I do not believe that Al Gore's statements reflected a mainstream position of climate scientists that the arctic would be ice free in the summer of 2013.
Joegoze,
The Gore quote you have produced is a nuanced statement that correctly states that scientists at the time projected the end of Arctic Sea ice between 7-22+ years. Your characterization of it claiming 2013 as a concrete date is false. You have only two quotes remaining.
Paul Beckwith is "Paul Beckwith is a Ph.D. student with the laboratory for paleoclimatology and climatology, department of geography, University of Ottawa.", hardly an important scientist or promenent like Senator Inhofe. He states in his post that he is an outlier. You have one quote.
The Kerry quote applies the minimum scientific estimate to a subject with a large range of projections and Kerry is in error. You have one quote against a half dozen for promenent deniers who claim the greenhouse effect does not occur.
If all you can find is a single politician who has misapplied a scientific study you have little to stand on. You have personally misquoted Al Gore so if we subtract one to compensate for your misquote you have no quotes left!
michael sweet:
You are making my point, all those are irrelavent people making irrelavent comments. Does not represent mainstream thinking in the science community. I am saying Prominance does not equal valid scientific argument.
[JH] This discussion has gone off-topic. Let's end it here. Cancel that.
joeygoze #27 The posting doesn't assert that a majority of people that question man's contribution to the greenhouse effect also do not believe in a greenhouse effect at ALL. I agree with you that people shouldn't make unsubstantiated claims and talk nonsense, though I gather some of those listed here are American celebrities or politicians so it seems like a totally hopeless hope in those cases. This "John Kerry" fellow has destroyed my piece of mind because I'd heard before it was a threat to healthy existence of all mammal & reptile life on Earth but I hadn't realized it's escalated to being a threat "— yes — even to American national security."
joeygoze - Kerry wasn't just giving an unsupported opinion regarding national security, either. Top U.S. Admiral: Climate Change Biggest Threat.
Other notes - claims that the greenhouse effect doesn't exist are, sadly, neither rational nor rare. They come up time and time again on pseudoskeptic blogs. And as to your quotes about 2013? You appear to have overlooked the qualifiers, such as in "...it could happen in as little as 7 years", not a sole prediction of that single year, and as such you are presenting a strawman argument.
KR @31, your link to a "pseudoskeptic blog" is to Jonova's explanation that greenhouse gases warm the Earth, not directly, but merely be retarding the escape of heat. A point on which she is correct. On the other hand, Joe Postma clearly argues in comments (and I strongly suspect others do to) that there is no greenhouse effect. It is probably best to clarrify these points, and even to acknowledge that JoNova is on the side of science on this point (if few others).
joeygoze@27,@29
You haven't answered my questions @24.
Instead, you're now making incorrect claims that this article equates those who deny FGE with those who deny that humans are responsible for AGW (it does not, debunked by grindupBaker@30).
Further, you're now redefining your objections to this article because it "does not represent mainstream thinking in the science community". I disagree. The purpose of his blog is not to follow "mainstream thinking in the science community" (as you would prefer), but to "Explaining climate change science & rebutting global warming misinformation" as you can read in the home page.
So I renew my question: what's the purpose of your objection? Reasonable and responsible people, do explain all basic aspects of life to their descendants (e.g. children), despite the fact that those basics are not "mainstream thinking in the science community". By analogy, this blog explains the basics of climate science to the bloggers who have not learnt them or have learnet them wrong from denialosphere. Why are you denying the very reason for SkS existence? What is the ethical reason behind that denial of yours?
Joeygoze,
It is not clear what you are trying to argue. You seem to be saying that we need to compare only those who you consider to be mainstream deniers to mainstream climate scientists. You claim that US Senators, popular denier blogs and the Prime Minister of Australia do not represent denier opinion.
You have misquoted Al Gore, found an expreme post from a low level scientist, and an instance where a non scientist made stronger claims than are justified scientificly. You have insisted on claiming Gore said that the ice would melt in 2013 when your quote does not support that claim, so you provide an instance as bad as Kerry. Claiming a blog post by Paul Beckwith is as important as public statements from Tony Abbott is absurd.
Deniers say that the greenhouse effect does not exist all the time. It is a waste of time to debate with someone who claims that is not a common argument when several examples have been provided.
UBrew12 @ #13
I don't wish to dismiss your analogy out of hand - because analogies can be useful in explaining abstract concepts to laypeople - but your dye in a swimming pool explanation is almost, but not quite, entirely incorrect in demonstrating the effect of CO2 concentrations in the atmosphere.
The pool dye advertised (apart from its almost complete lack of usefulness in anything other than some trivial First World sense) has none of the properties of CO2, does not behave in the atmposphere as CO2 does, nor does it emulate any of the physics necessary to explain CO2 influence. Suffice it to say - without boring all in the know - that explanations of the electromagnetic energy transfer and blocking mechanisms of CO2, its mixing levels at given temperatures and energy levels, and its raison d'etre in the equations of a myriad thermodynamic equations (such as the Stefan-Boltzmann), amongst other things, using an analogy that only shows a superficial resemblance to reality is misleading and unhelpful.
We need to argue against the deniers with objective, scientifically robust and valid empirical proof. Otherwise we risk dissembling into argument entropy where each side attempts to explain hyper-complex realms with simpler and simpler examples that results in a complete loss of any form of epistemological basis...And I, for one, would rather 'lose' an argument to a denier than relinquish that.
[PW] To all:
I'm as vociferous a fan of 'outing' those who dismiss the science of climate change as any, but let's all *drop* the use of the d-word; if you feel compelled, use 'dismissive,' or 'pseudoskeptic.' It only gives the dismissives ammo and it doesn't truly reflect the reality. There are true skeptics--pretty much 100% of all scientists--and pseudoskeptics, those who have little to no training in what they are dismissing. Not only do we need to "argue against the deniers with objective, scientifically robust and valid empirical proof.", we also need to do it as respectfully as we can.