Ocean Acidification Is Fatal To Fish
Posted on 22 December 2011 by Rob Painting
Fossil fuel-burning is acidifying the oceans and, up until recently, it has generally been thought that the greatest risk posed by ocean acidification was the change to seawater carbon chemistry. This is because rising levels of atmospheric carbon dioxide reduce the concentration of seawater carbonate ions, a vital building-block in the shells and skeletons of many marine life. Fish were not thought to be at direct risk from acidification, because they clearly don't build shells, and were considered to have well-developed physical mechanisms to tolerate falling pH (acidification).
Several studies published in the journal Nature Climate Change, Baumann (2011) and Frommel (2011), indicate that this might not be the case. Fish may, in fact, be seriously threatened by ocean acidification. Although adult fish seem well-equipped to deal with low pH waters, or higher levels of CO2 in seawater, their egg and larval life stages, a typically vulnerable time for all marine life, may not be so fortunate.
Baumann (2011) show that larvae survival in one fish species drops with increased levels of CO2 (figure 1). Survival rates plummeting some 75% under a scenario with 1000ppm (parts per million) of atmospheric CO2. And Frommel (2011) discovered considerable tissue damage and necrosis (dead tissue) in fish larvae of another species exposed to higher levels of CO2 than the present day. In the high CO2 experiments, this damage to internal organs was so extensive it lead to the death of afflicted larvae. Each of these studies are discussed in detail below.
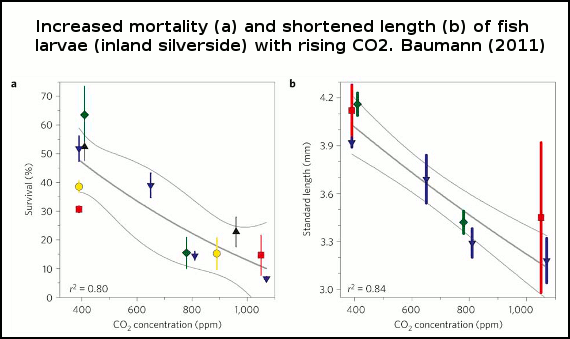
Figure 1 -Survival rates of larvae with increasing CO2. Thin grey lines=error bars (95% confidence intervals). Experiment 1, red squares; experiment 2, blue down triangles; experiment 3, green diamonds; experiment 4, yellow circles; experiment 5, black up triangles. Points represent means±1 standard deviation (a spread of measurements deviating from the average).
Ocean acidity hotspots
It's important to point out that some regions of the oceans, especially coastal waters, can seasonally experience extremely acidified waters, with much lower pH than the global average. For example, due to upwelling over the warmer months, Kiel Fjord in the Western Baltic Sea (Germany) can see prolonged periods of acidification so low that it is equivalent to atmospheric CO2 levels of 2300 ppm. So this needs to be kept in mind when the reading the levels of atmospheric CO2 discussed in the following studies. High levels of ocean acidification are already occurring in some areas of the ocean where fish spawning fish takes place.
One fish, two fish, shorter fish, dead fish
The inland silverside is an ecologically important small fish native to the Atlantic coast of North America, and is found in estuaries up-and-down the coast. It can also be found in acidified freshwater environments, and humans have even successfully transplanted them into land-locked freshwater systems and lakes, so they clearly can adapt to low pH (acidified) water.
Baumann (2011) conducted a series of experiments where CO2 was bubbled into seawater containing the newly-fertilized eggs of the inland silverside. Larvae were exposed to levels of acidification representative of modern-day (390-400ppm,) right up to projected late 21st century (900-1100ppm) atmospheric CO2 levels. After a week the surviving larvae were counted and measured.
In each of the 5 experiments both the larvae body length and survival rates declined as seawater became more acidified (figure 1), and there were also more deformed larvae (figure 2).
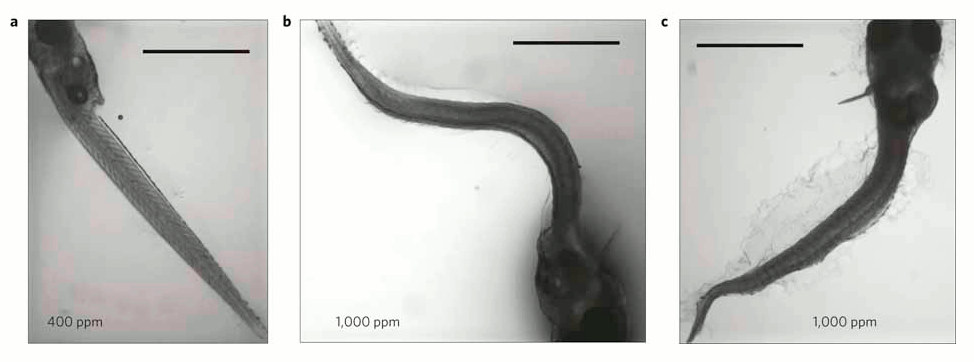
Figure 2- Larvae with curved or curled bodies were significantly more common at increased (b,c) when compared with control (a) CO2 levels. Scale bar=1 mm.
At 1000 ppm, survival plunged some 75% and larval body length 18%. The shorter length indicates a slower rate of growth, a problem for larvae because the longer the time they spend growing in their larval stage, the higher the rate of predation by plankton-eating fish and other critters. In other words, slower growth and larval deformities will likely translate into even lower survival rates in the wild, compounding the death rate of these larvae as ocean acidification increases.
The cause of this slow growth, deformation and decline in survival rates in the experiments is not known, but given that inland silverside can grow and thrive in more acidified freshwater, this suggests something other than pH. Baumann (2011) suggest this may be related to high CO2 levels, or even the carbonate chemistry of the water.
The authors speculated that the negative response may have been related to the susceptibility of very early larval development, the egg embryo, so carried out another experiment, After growing in seawater equivalent to 410 ppm for 5 days, fertilized eggs were exposed to 780ppm once they hatched. Survival rates only slightly dropped in this treatment versus the control (at constant 410 ppm), and survival was much higher than larvae exposed to 780 ppm the entire time, suggesting that it's the eggs themselves that may be vulnerable to ocean acidification.
Acidic oceans severely damage internal organs in fish larvae
Frommel (2011) was another experiment carried out on fish larvae, this time to focus was on Atlantic cod, a fish widely distributed throughout the North Atlantic Ocean, and one listed as vulnerable on the IUCN Red List of threatened species. This is the same species that was decimated by overfishing in the late 20th century, an activity which lead to the collapse of 6 populations off the Canadian coastline.
The cod larvae for this experiment were taken from a population off Norway, and were exposed to 3 levels of seawater acidity equivalent to 380 ppm (the control), 1800 ppm (medium) and 4200 ppm (high). Although these last two scenarios may seem very high, Kiel Fjord (noted earlier) is very near to Baltic cod spawning grounds and already sees levels above the equivalent of 1800ppm, and is likely to endure acidification comparable to around 4200 ppm with a doubling of atmsopheric CO2 (560 ppm). So the experiments are directly relevant to current and near-future conditions in which Atlantic cod spawn.
Cod larvae were reared for 7 weeks in large outdoor cylinders called mesocosms. The authors did not directly test for mortality (death rate), but instead looked for changes in tissue health. They discovered that in the early stages of development, the first 25 days after hatching was a critical time. Newly-hatched larvae lack fully-functioning gills, which is the primary organ for helping regulate internal pH balance and CO2 build up in tissue. In this early development stage the larvae are essentially at the mercy of the surrounding seawater pH, and are therefore very vulnerable. In the experiment, larvae suffered extensive damage to vital organs with rising acidity, and those larvae with extreme malformations to internal organs and widespead dead tissue, obviously died from these deformities. See figure 3.
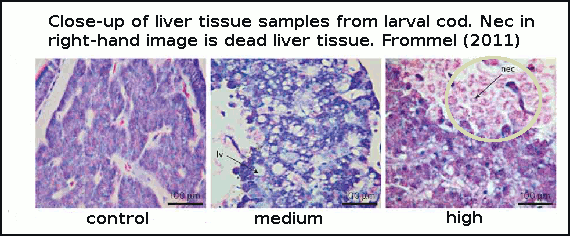
Figure 3 - histological sections (tissue samples under microscope magnification) of cod larvae liver at increasing CO2. Control=380ppm, medium=1800ppm and high=4200ppm.
The effect of the three acidity scenarios on larval tissue are is shown in figure 4. As acidity increases, so too does the amount of tissue damage.
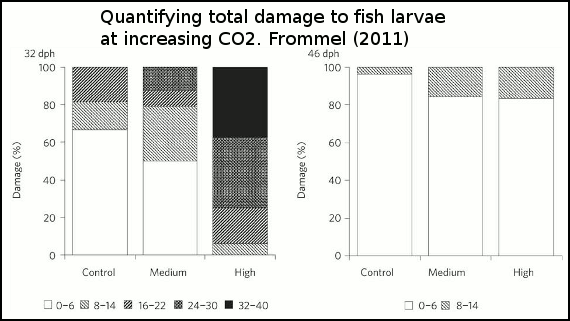
Figure 4 - Percentage of larvae exhibiting different degrees of total damage at 32 and 46 days after hatching and three different treatment levels (control, medium and high). The damage is shown as five levels with normal as white bars, and shading increasing with increasing severity of damage. Control=380ppm, medium=1800ppm and high=4200ppm.
In the summary of their paper the authors conclude:
"Although we did not directly test for mortality rates, our data on severe tissue damage suggest that ocean acidification will negatively impact the recruitment of mass-spawning fishes because of enhanced mortality rates."
Fish flunk the acid test too
To sum up:
- The Baumann (2011) and Frommel (2011) studies are surprising in that they reveal that rather than being impervious to the effects of ocean acidification, as earlier believed, higher levels of acidification are actually fatal to fish in their early life stages (egg/larval form).
- Both the severity of tissue damage to major internal organs, and the mortality rate in fish larvae increase along with rising levels of CO2.
- The high CO2 scenarios in the experiments are representative of both current day and near-future conditions found in some regions where these fish spawn. So the results suggest ocean acidification may be having an impact on these species today.
- Further tests will be necessary to determine how widespread this egg/larval susceptibility is among fish species, but if the results of these studies are an indication of the harm ocean acidification causes to fish life in general, then along with overfishing, pollution and habitat contraction (a consequence of warming seawater and ocean acidification) we may begin to see major fish stocks collapse.
Related SkS posts: OA not OK series, Ocean Acidification: Corrosive waters arrive in the Bering Sea, Acidification:Oceans past, present & yet to come, Ocean acidification: Some Winners, Many Losers, Ocean acidification: global warming's evil twin and Great Barrier Reef Part 3: Acidification, Warming, and Past Coral Survival.































 Arguments
Arguments






























[DB] You are missing the fundamental point that the rate of change is the issue with today's human-induced climate change. The rate of CO2 injection into the atmosphere is more than 10x that which occurred during the PETM, the most comparable period in Earth's history. And, as you point out, CO2 is correlated with global warming (and cooling).
Please read the CO2 Currently Rising Faster Than The PETM Extinction Event thread.
[DB] See the response to your previous statement.
What's his next experiment, setting fire to a cat?[DB] "Obviously, when conditions change, some species die, some flourish."
Your concerns are better addressed on the Climate's changed before thread. Please avoid statements such as your last, which adds nothing to the dialogue and is largely interpretable as trolling.
[DB] "What we need to be doing is to improve our models"
Please take your concerns to the Models are unreliable thread.
[DB] "skeptic-deniers can point to the spikes that occurred during the pleistocene period as being greater than what happened today"
You mean as you are doing now? Your concerns are better addressed in the Climate's changed before thread. And you are incorrect for multiple reasons. Please read the referenced thread and place any remaining concerns there.
[DB] "Please don't put me in the same bucket as those nutcases."
I do not. However, repeating a meme gives it undeserved life. Perhaps a slight rephrasing of your statements would be more effective:
"One thing that I've heard repeated in some circles is that the temperature and CO2 spikes that occurred during the Pleistocene period were greater than what is happening today. Can someone help me understand why this statement isn't true or relevant to today?"
I think the experiments above were pointless, who didnt know if you keep seafish in almost neutral water they would die and produce deoformed fry especially when they havent had 96 years to adapt to the change?
Vonnegut, even if something looks obvious, it still needs to be confirmed via the scientific method. There is a variety of information discoverable well beyond the basic confirmation or rejection of the primary hypothesis.
To me, it's obvious that anthropogenic global warming is occurring and that humans are overhwlemingly responsible for the trend of the last 50 years. So why are we paying these scientists to work out the details. How pointless!
vonnegut, how could fish adapt to the change in as little as only 96 years?
The inland silverside is a fish that lives in estuaries and freshwater, this to me at least makes it surprising that OA proved to be such a problem for them. The fact that the work was published in a journal suggests that the outcome was non-obvious and the experiments were not pointless.
I should point out again that your posting style is not going to work well here, dismissing scientific research as pointless without paing attention to details (such as the natural habitat of the inland silverside) does not give confidence.
@41 the same way they adapt to changes in Ph and temp every day? bit by bit.
Take a fish from 58 degrees and drop in 80f water and it will die within hours if you do small increases over a few weeks they can survive.
Fair point ,noted.
vonnegut wrote "the same way they adapt to changes in Ph and temp every day? bit by bit."
This is the same discussion we had on the other thread, just because X is tolerant to short term variations in Y does not imply that X is tolerant of long term changes in Y of similar magnitude. As I pointed out on the other thread, UK native fauna and flora can quite happily adapt to a diurnal and seasonal temperature changes of 15 degrees C, not much of it could adapt to a permanent change of 15 degrees C.
I guess asking how the coelacanth made it to modern day is futile :)
@43 youre suggesting that its harder to adapt over time than it is day by day?
0.1 ph rise is nowhere near the same as a 15 degree rise in mean temp, oddly enough using that analogy has made me think of Inuit, wonder what their average mean is?
vonnegut wrote "youre suggesting that its harder to adapt over time than it is day by day?"
No, I am saying that oscillatory vairiablity is easier to adapt to than long term effectively permanent change. I would have thought that was obvious from my contrasting "diurnal and seasonal temperature changes" with "permanent change".
I notice that on this thread again you are ignoring the example I gave. Do you think that UK native flora and fauna could adapt to a permanent drop of 15 degrees C in temperature, yes or no?
Second thoughts, I have gone back to thinking you are just trolling. You have no basis for saying that "0.1 ph rise is nowhere near the same as a 15 degree rise in mean tem". pHs and temperatures are not directly comparable without reference to the effects they have on the environment. UK flora and fauna has no problem with a 15 degree C daily variability, fish have no problem with a daily or seasonal 0.1 change in pH. That doesn't mean that fish can cope with a permanent change of 0.1 in pH and more than UK flora and fauna can deal with a permanent change of 15 degrees C in mean temperature.
The real slice of baloney though is the comment about the inuit. Sure inuit can adapt, but you may have noticed that the flora and fauna they encounter is rather different to the flora and fauna you are likely to encounter in the U.K. Do you think that just possibly the difference in mean temperature might be the reason?
Sorry, life is too short for this sort of persiflage when genuine discussion of the science is so much more interesting.
Ive been searching for info on the ph range of the Menidia beryllina to no avail (anyone?)
also wasnt the experiment rather futile as the fish may not have spawned in such low ph water?
Well Dikran youre entitled to your opinion. Yes a permanent drop of 15 degrees would affect many creatures, I just dont think a small drop in ph will, thats all.
I thank you for your input so far.
Vonnegut, you seem to be searching for something that allows you to say, "gotcha!" and then walk away. If you were actually trying to understand how it all works, you'd lose the "you're so dumb" attitude and read over both the entire OA is not OK series and some of the more comprehensive studies, starting with Honisch et al. 2012. You'd then say, "ok, this is how I understand it . . . am I right?"
Instead, you're saying, "Ok, this is how I understand it, and I don't really care for your amateurish opinion on my understanding. It's clear that this is not happening or is not a problem. No, I don't need evidence. Or, rather, I need only need evidence to the extent that the evidence supports my pre-existing opinion."
I'll also point out that it's fine to play devil's advocate (e.g. "but what about X?"), but it's not ok to attach subtext that argues that scientists don't know what they're talking about and/or are engaged in fraud. You can draw those conclusions once you've read the existing research and have evidence that fraud is taking place. Until then, lose the accusatory rhetoric.